|
The electronic configuration of cobalt is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 7 4s 2. What is the electronic configuration of cobalt? There are nine valence electrons in an atom of Cobalt.The electron configuration of Cobalt is: 1s 2 2s 2 2p 6 3s 2 3p 6 3d 7 4s 2.Read: What are Valence Electrons? Key Takeaways In general, cobalt is a versatile element with a range of valencies and oxidation states, making it useful in many different applications. This is less common than the +2 oxidation state, but it is still important in some applications, such as in the production of cobalt-based catalysts.Ĭobalt can also form complex ions with various ligands, resulting in a range of oxidation states and valencies. Cobalt(II) is a common oxidation state for cobalt in many compounds, such as cobalt chloride (CoCl2) and cobalt sulfate (CoSO4).Ĭobalt can also have a valency of +3 in some compounds, where it loses three electrons to form a cation with a 3+ charge. This is because cobalt has two valence electrons in its outermost shell (in the 4s orbital) which it can lose to form a stable electronic configuration, similar to the noble gas configuration of argon. In its most common ionic form, cobalt has a valency of +2, meaning it loses two electrons to form a cation with a 2+ charge. The valency of cobalt can vary depending on the chemical compound it is present in. In the case of cobalt, the presence of seven valence electrons in the 3d orbital can contribute to its ability to form complex compounds with other elements and ions. The number of valence electrons in an atom is often used to predict its reactivity and its ability to form chemical bonds with other atoms. The electron configuration of cobalt is 3d 7 4s 2, indicating that there are two valence electrons in the 4s orbital and seven valence electrons in the 3d orbital. In the case of cobalt, the valence electrons are those in the 4s and 3d orbitals. Valence electrons are the outermost electrons in an atom that are involved in chemical reactions and bonding with other atoms. 
Valence Electrons in CobaltĬobalt has a total of nine valence electrons. The third shell contains a total of 17 electrons, with two electrons in the 3s orbital, six electrons in the 3p orbital, and the remaining nine electrons in the 3d orbital. The second shell contains eight electrons, which occupy the 2s and 2p orbitals. The first shell of cobalt contains two electrons, which occupy the 1s orbital. 
The electron distribution in the shells of a cobalt atom is as follows: Distribution of Electrons in Shell in Cobalt Atom This shorthand notation is used to indicate that the 18 electrons in the argon configuration are not involved in chemical reactions, and only the valence electrons in the 3d and 4s orbitals participate in bonding.Ĭhlorine Electron Configuration 3. The electronic configuration of cobalt can also be written in the shorthand notation as 3d 7 4s 2, where represents the electronic configuration of the noble gas argon (which has 18 electrons). The remaining seven electrons are distributed among the 3d and 4s orbitals, with three electrons in the 3d sub-level and two electrons in the 4s sub-level. The next ten electrons occupy the 3s and 3p orbitals. In the electronic configuration of cobalt, the first two electrons occupy the 1s orbital, the next two electrons occupy the 2s orbital, and the following six electrons occupy the 2p orbital. 
The electronic configuration represents how those 27 electrons are distributed among the different energy levels and sub-levels of the cobalt atom. The electronic configuration of cobalt is:Ĭobalt has an atomic number of 27, which means it has 27 electrons. 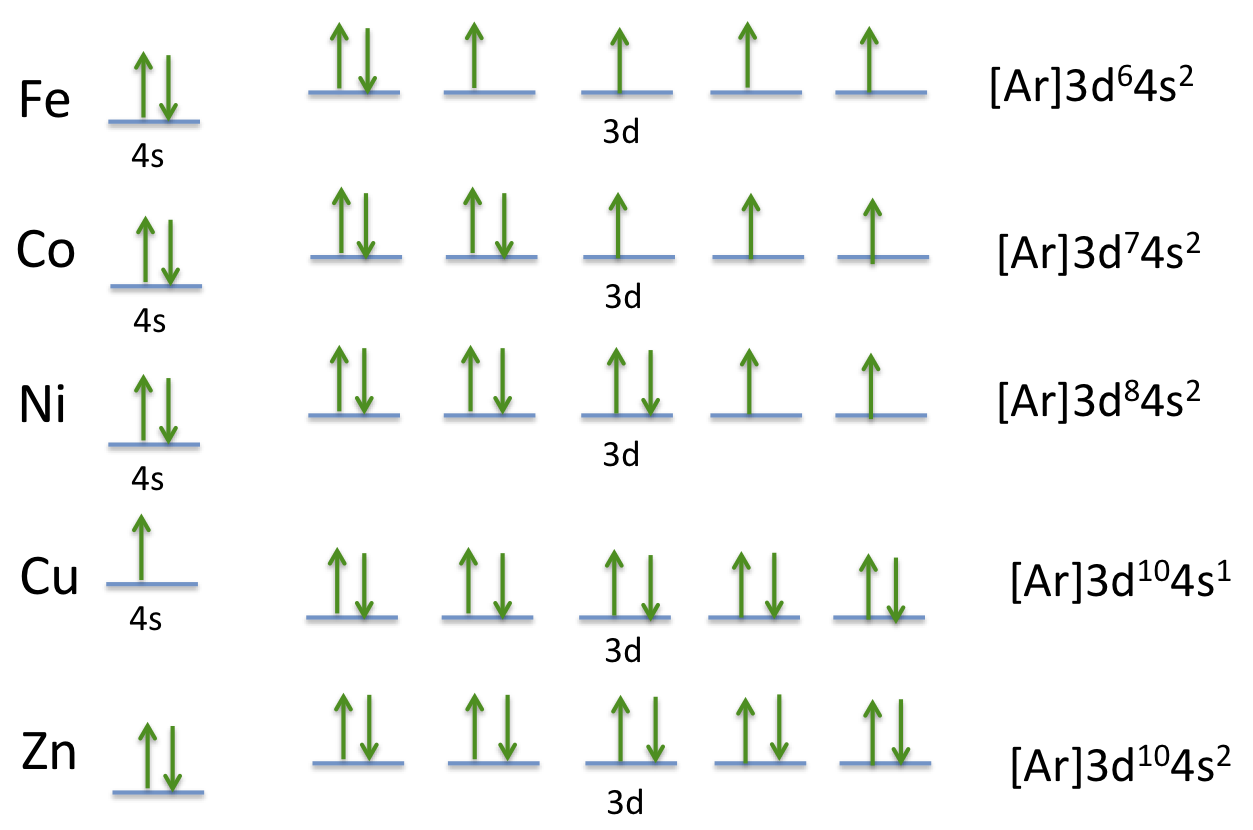
The largest producers of cobalt are the Democratic Republic of the Congo, Zambia, and Australia. It is also used as a pigment in ceramics and glass, and as a catalyst in chemical reactions.Ĭobalt is found in the Earth’s crust, usually in combination with other metals, such as copper, nickel, and arsenic. Cobalt is also used in various industrial and technological applications, such as in the production of superalloys for use in jet engines and gas turbines, in the manufacture of magnets and magnetic storage media, and in the production of rechargeable batteries.Ĭobalt has a high melting point and is relatively resistant to corrosion, making it a useful material for high-temperature applications. It is used in small amounts as a cofactor in enzymes and proteins, including vitamin B12. It is a hard, gray metal that belongs to the group of transition metals in the periodic table.Ĭobalt is an essential trace element for many living organisms, including humans. Cobalt is a chemical element with the symbol Co and atomic number 27.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
